 The study was conducted in the Community Holistic Health Center, Hillsborough, NC, in 1995-1996. It consisted of following up 200 subjects who used their own devices for at least two weeks. 126 of them filled out the questionnaires which were collected and analyzed. Among them, 105 reported that they used the device for various specific health problems The five most frequently reported conditions were the following (starting with the most frequent): stress, pain, muscle spasms, mood swings and insomnia.
The study was conducted in the Community Holistic Health Center, Hillsborough, NC, in 1995-1996. It consisted of following up 200 subjects who used their own devices for at least two weeks. 126 of them filled out the questionnaires which were collected and analyzed. Among them, 105 reported that they used the device for various specific health problems The five most frequently reported conditions were the following (starting with the most frequent): stress, pain, muscle spasms, mood swings and insomnia.
Results
One or more positive effects of the device on the conditions specified by subjects were reported by 99 of the 105 participants (94%) with the above specific health problems. To obtain significant number of cases, we included in the questionnaire 5 health conditions to be estimated by all the participants, including those with no particular health problems. Out of the 21 participants who reported that they tested the method while having no particular health problems, 13 nevertheless reported one or more positive effects of the ASHA*. The only adverse effect is some initial discomfort which disappears after a few treatments. In fact, one of the questions in our research questionnaire was the following: “What was your comfort level the first time you laid on the ASHA*? The fifth time?” During the first use 46% felt very uncomfortable and 42% uncomfortable. However, by the fifth use, the picture has reversed: 52.5% felt very comfortable and 36.5% – comfortable.
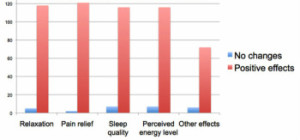
One of the questions of the questionnaire was the following: “Did you experience any positive effects using the ASHA* in the following categories: relaxation, pain relief, sleep quality, energy level, allergies?” Out of 126 subjects, 98% reported pain relief, 96% reported relaxation, 94% reported improvement in the quality of sleep, and 81% reported an increase in energy level.
Source: Zilberter T, Roman J. Reflexo-Therapy with Mechanical Skin Stimulation: Pilot Study. International Symposium on Integrative Medicine. New York: Omega Institute, 1999 Download PDF
——————————–
* The device used in the study was an earlier version of ASHA manufactured in the US in 1993-2000 under the name of Panacea

